March 20, 2024 — Since its introduction, the Shingrix vaccine has been hailed as a breakthrough in the prevention of shingles.
But the path to widespread adoption is littered with obstacles, from pandemic-related disruptions to insurance complexities. The impact is reflected in the number of vaccines. According to one study, only about 30% of adults eligible for Shingrix have received the shot. Government Accountability Board 2022 Report.
Medical experts support its use, emphasizing its unparalleled effectiveness in preventing painful conditions.
„Herpes zoster is a leading cause of disability around the world, and a fast-acting and highly effective vaccine against shingles is now in development,“ he said. Kenneth Consija, MD, specialist at the Cleveland Clinic Geriatrics Center. “However, since its release, it has remained a very muddy landscape for various reasons, which complicates matters.”
Shingrix, a two-dose shingles vaccine that the FDA approved in 2017, was touted as a much more effective alternative than its predecessor, Zostavax.
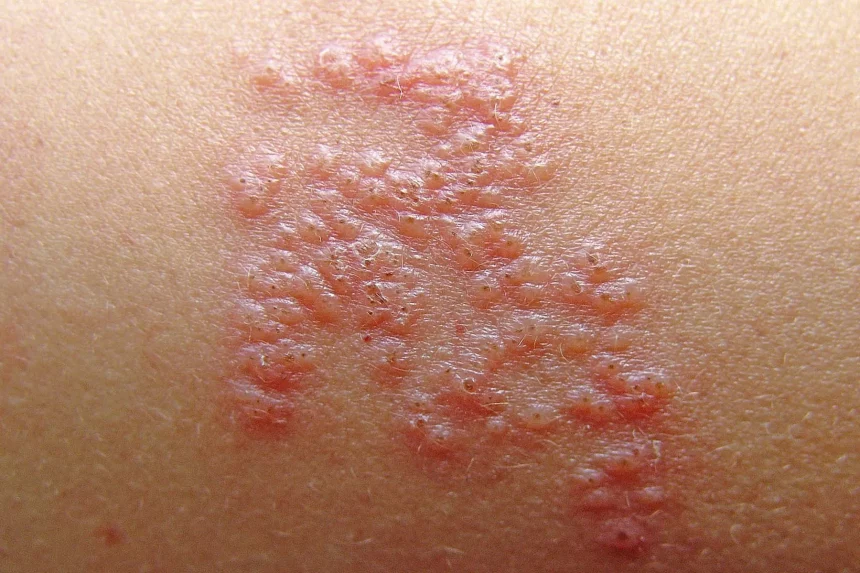
of CDC It is estimated that approximately 1 in 3 people in the United States will develop shingles. About 10% to 18% of them experience postherpetic neuralgia (PHN), which is nerve pain, or burning pain in the nerves and skin. It can last for years even after the rash has subsided. Other potential complications include serious eye, heart, and nervous system problems, and even death.
It can also increase your risk of cardiovascular events such as heart attack and stroke by nearly 30%, according to a study published in . American Heart Association Journal.
While Zostavax reduced the risk of shingles by 51%, Shingrix's effectiveness rose to 97% in people aged 50 to 69 and 91% in people 70 and older. In addition, the preventive effect on postherpetic neuralgia is approximately 90% in all age groups, whereas the preventive effect on postherpetic neuralgia is approximately 90% in all age groups.Zostavax.
But in 2017, the concept of a second vaccination was daunting for many, and out-of-pocket costs became even more complicated.
„This was long before COVID-19, and many people didn't even know what the word 'booster' meant at that point,“ Consilia said.
High prices were a major hindrance. Many Medicare Part D prescription drug plans required a copayment for the shingles vaccine.according to 2019 Report to Congress Vaccines by Medicare Payment Advisory CommissionIf Medicare recipients don't meet their deductible, the two required shots could cost more than $400.
Things changed in Congress in 2023. Eliminating the cost burden for vaccines Approved by the CDC's Advisory Committee on Adult Immunization Practices. This policy applies regardless of whether people have drug coverage through a Part D or Medicare Advantage plan that includes the shingles vaccine.
“The cost was high, Out of stock soon'' said Dr. Tina Ardon, a family medicine physician at the Mayo Clinic in Jacksonville, Florida. „Some people only got one shot.“
Concerns about potential side effects were an additional deterrent. According to the CDC, the vaccine can cause patients to experience arm pain, redness and swelling at the injection site, fatigue, muscle pain, headache, chills, fever, abdominal pain and nausea. These side effects may last for 2-3 days.
The coronavirus pandemic has added further complexity, fueling vaccine hesitancy and reducing uptake rates. Among adults who receive commercial news, Claims regarding vaccination According to the report, December 2020 saw a 15% decrease compared to December 2019, and April 2021 saw a 62% decrease compared to April 2019.
„Do vaccines work? Yes, they work incredibly well,“ said Timothy Brewer, MD. Professor of Medicine, Department of Infectious Diseases, David Geffen School of Medicine, University of California, Los Angeles. „It's really great that we have a vaccine available for this population and that it's working. We're hopeful that uptake will improve.“